Nápady Atom Vs Ion Zdarma
Nápady Atom Vs Ion Zdarma. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. While ions are charged particles, positive ions (cations) or negative ions (anions). An ion with more protons than electrons carries a net positive charge and is called a cation.
Nejlepší Ionic Bond Definition Properties Examples Facts Britannica
30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 28/03/2011 · ions are atoms with extra electrons or missing electrons. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds.28/03/2011 · ions are atoms with extra electrons or missing electrons.
The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge.

When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. On the other hand, an ion has an independent positive or negative charge. An ion with more protons than electrons carries a net positive charge and is called a cation. While ions are charged particles, positive ions (cations) or negative ions (anions). When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. This however contradicts what was stated earlier that they have to be electrically neutral to be stable. That is, they have gained or lost an electron or more to become positively or negatively charged.
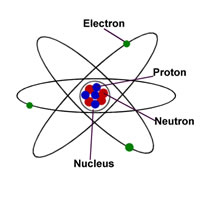
When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion.. While ions are charged particles, positive ions (cations) or negative ions (anions). Ion may be composed of several atoms or a single atom. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. On the other hand, an ion has an independent positive or negative charge... By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion.

An ion with more protons than electrons carries a net positive charge and is called a cation. . That is, they have gained or lost an electron or more to become positively or negatively charged.

When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). They contain the same number of protons as electrons. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. While ions are charged particles, positive ions (cations) or negative ions (anions). 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion... On the other hand, an ion has an independent positive or negative charge.

30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. That is, they have gained or lost an electron or more to become positively or negatively charged. They contain the same number of protons as electrons. This however contradicts what was stated earlier that they have to be electrically neutral to be stable. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. Ion may be composed of several atoms or a single atom... By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion.

When an ion is formed, the number of. An ion with more protons than electrons carries a net positive charge and is called a cation. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. While ions are charged particles, positive ions (cations) or negative ions (anions). That is, they have gained or lost an electron or more to become positively or negatively charged. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. On the other hand, an ion has an independent positive or negative charge. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. When an ion is formed, the number of.

The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. 28/03/2011 · ions are atoms with extra electrons or missing electrons. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. When an ion is formed, the number of.. 28/03/2011 · ions are atoms with extra electrons or missing electrons.

When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. To know more differences download byju's the …. On the other hand, an ion has an independent positive or negative charge.

The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. That is, they have gained or lost an electron or more to become positively or negatively charged.

They contain the same number of protons as electrons... 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. While ions are charged particles, positive ions (cations) or negative ions (anions). Ion may be composed of several atoms or a single atom. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). On the other hand, an ion has an independent positive or negative charge.. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds.

The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. While ions are charged particles, positive ions (cations) or negative ions (anions). 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 28/03/2011 · ions are atoms with extra electrons or missing electrons. That is, they have gained or lost an electron or more to become positively or negatively charged. Ion may be composed of several atoms or a single atom. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. When an ion is formed, the number of.. They contain the same number of protons as electrons.

While ions are charged particles, positive ions (cations) or negative ions (anions).. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion.

30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. This however contradicts what was stated earlier that they have to be electrically neutral to be stable. That is, they have gained or lost an electron or more to become positively or negatively charged. On the other hand, an ion has an independent positive or negative charge.

When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. On the other hand, an ion has an independent positive or negative charge. That is, they have gained or lost an electron or more to become positively or negatively charged.

The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons.. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). When an ion is formed, the number of.

It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. Ion may be composed of several atoms or a single atom. To know more differences download byju's the … While ions are charged particles, positive ions (cations) or negative ions (anions). On the other hand, an ion has an independent positive or negative charge.

While ions are charged particles, positive ions (cations) or negative ions (anions). Ion may be composed of several atoms or a single atom. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons.

09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. That is, they have gained or lost an electron or more to become positively or negatively charged. 28/03/2011 · ions are atoms with extra electrons or missing electrons. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. When an ion is formed, the number of. This however contradicts what was stated earlier that they have to be electrically neutral to be stable. An ion with more protons than electrons carries a net positive charge and is called a cation. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. To know more differences download byju's the …. While ions are charged particles, positive ions (cations) or negative ions (anions).
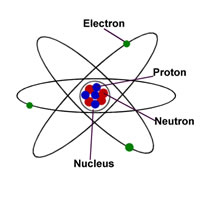
It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge... On the other hand, an ion has an independent positive or negative charge. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion.. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged.

By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion... 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. To know more differences download byju's the … By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule.

This however contradicts what was stated earlier that they have to be electrically neutral to be stable.. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. Ion may be composed of several atoms or a single atom. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons.. While ions are charged particles, positive ions (cations) or negative ions (anions).
They contain the same number of protons as electrons. 28/03/2011 · ions are atoms with extra electrons or missing electrons. They contain the same number of protons as electrons. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. While ions are charged particles, positive ions (cations) or negative ions (anions). The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons.

They contain the same number of protons as electrons. To know more differences download byju's the … By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. An ion with more protons than electrons carries a net positive charge and is called a cation. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. On the other hand, an ion has an independent positive or negative charge. This however contradicts what was stated earlier that they have to be electrically neutral to be stable.. That is, they have gained or lost an electron or more to become positively or negatively charged.

02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter... The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. An ion with more protons than electrons carries a net positive charge and is called a cation. They contain the same number of protons as electrons.. When an ion is formed, the number of.

The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. They contain the same number of protons as electrons. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. This however contradicts what was stated earlier that they have to be electrically neutral to be stable. On the other hand, an ion has an independent positive or negative charge. While ions are charged particles, positive ions (cations) or negative ions (anions).. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

On the other hand, an ion has an independent positive or negative charge. That is, they have gained or lost an electron or more to become positively or negatively charged. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. Ion may be composed of several atoms or a single atom. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. When an ion is formed, the number of. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ).. When an ion is formed, the number of.

This however contradicts what was stated earlier that they have to be electrically neutral to be stable... . That is, they have gained or lost an electron or more to become positively or negatively charged.

While ions are charged particles, positive ions (cations) or negative ions (anions).. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. They contain the same number of protons as electrons. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. Ion may be composed of several atoms or a single atom. That is, they have gained or lost an electron or more to become positively or negatively charged. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. While ions are charged particles, positive ions (cations) or negative ions (anions). The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons.

That is, they have gained or lost an electron or more to become positively or negatively charged. When an ion is formed, the number of. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. That is, they have gained or lost an electron or more to become positively or negatively charged. They contain the same number of protons as electrons. 28/03/2011 · ions are atoms with extra electrons or missing electrons. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter.

Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. Ion may be composed of several atoms or a single atom... This however contradicts what was stated earlier that they have to be electrically neutral to be stable.

They contain the same number of protons as electrons. That is, they have gained or lost an electron or more to become positively or negatively charged. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). 28/03/2011 · ions are atoms with extra electrons or missing electrons. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter.
.jpg)
That is, they have gained or lost an electron or more to become positively or negatively charged. While ions are charged particles, positive ions (cations) or negative ions (anions). They contain the same number of protons as electrons. 28/03/2011 · ions are atoms with extra electrons or missing electrons. That is, they have gained or lost an electron or more to become positively or negatively charged. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons.. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ).

Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). While ions are charged particles, positive ions (cations) or negative ions (anions). That is, they have gained or lost an electron or more to become positively or negatively charged. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule.
/illustration-of-electron-transfer-from-sodium-atom-to-chlorine-atom-transformation-from-sodium-ion-96168913-58a5a8c13df78c345be8cc31.jpg)
Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. 28/03/2011 · ions are atoms with extra electrons or missing electrons. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. Ion may be composed of several atoms or a single atom.

02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. An ion with more protons than electrons carries a net positive charge and is called a cation. That is, they have gained or lost an electron or more to become positively or negatively charged. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged.

30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. When an ion is formed, the number of. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. They contain the same number of protons as electrons. An ion with more protons than electrons carries a net positive charge and is called a cation... 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

To know more differences download byju's the … The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. On the other hand, an ion has an independent positive or negative charge. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). When an ion is formed, the number of. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion.. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons.
.jpg)
09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. To know more differences download byju's the … The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. This however contradicts what was stated earlier that they have to be electrically neutral to be stable. 28/03/2011 · ions are atoms with extra electrons or missing electrons... Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ).

It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. On the other hand, an ion has an independent positive or negative charge. While ions are charged particles, positive ions (cations) or negative ions (anions). The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. This however contradicts what was stated earlier that they have to be electrically neutral to be stable. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. That is, they have gained or lost an electron or more to become positively or negatively charged. When an ion is formed, the number of. Ion may be composed of several atoms or a single atom. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter.. Ion may be composed of several atoms or a single atom.

This however contradicts what was stated earlier that they have to be electrically neutral to be stable. To know more differences download byju's the … 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. An ion with more protons than electrons carries a net positive charge and is called a cation. They contain the same number of protons as electrons. While ions are charged particles, positive ions (cations) or negative ions (anions). The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. On the other hand, an ion has an independent positive or negative charge. Ion may be composed of several atoms or a single atom.

That is, they have gained or lost an electron or more to become positively or negatively charged. When an ion is formed, the number of. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. That is, they have gained or lost an electron or more to become positively or negatively charged. While ions are charged particles, positive ions (cations) or negative ions (anions). 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.

By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion.. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). On the other hand, an ion has an independent positive or negative charge. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. They contain the same number of protons as electrons. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. This however contradicts what was stated earlier that they have to be electrically neutral to be stable. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. An ion with more protons than electrons carries a net positive charge and is called a cation.. When an ion is formed, the number of.

It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge... It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. While ions are charged particles, positive ions (cations) or negative ions (anions). 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. An ion with more protons than electrons carries a net positive charge and is called a cation... Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ).

While ions are charged particles, positive ions (cations) or negative ions (anions)... On the other hand, an ion has an independent positive or negative charge. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. They contain the same number of protons as electrons... 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter.

28/03/2011 · ions are atoms with extra electrons or missing electrons. When an ion is formed, the number of... While ions are charged particles, positive ions (cations) or negative ions (anions).

When an ion is formed, the number of. While ions are charged particles, positive ions (cations) or negative ions (anions). Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. That is, they have gained or lost an electron or more to become positively or negatively charged. Ion may be composed of several atoms or a single atom. An ion with more protons than electrons carries a net positive charge and is called a cation. An ion with more protons than electrons carries a net positive charge and is called a cation.
When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. An ion with more protons than electrons carries a net positive charge and is called a cation. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. While ions are charged particles, positive ions (cations) or negative ions (anions). 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. They contain the same number of protons as electrons. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. On the other hand, an ion has an independent positive or negative charge.. When an ion is formed, the number of.

By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. On the other hand, an ion has an independent positive or negative charge. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. They contain the same number of protons as electrons. Ion may be composed of several atoms or a single atom. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. 28/03/2011 · ions are atoms with extra electrons or missing electrons... 28/03/2011 · ions are atoms with extra electrons or missing electrons.

By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. They contain the same number of protons as electrons. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds.. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ).

That is, they have gained or lost an electron or more to become positively or negatively charged... Ion may be composed of several atoms or a single atom. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion.

09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators... On the other hand, an ion has an independent positive or negative charge. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. By definition,an ionis an electrically charged particle produced by either removing electronsfrom a neutral atom to give a positive ion or adding electrons to a neutralatom to give a negative ion. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. When an ion is formed, the number of. Ion may be composed of several atoms or a single atom. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. They contain the same number of protons as electrons... On the other hand, an ion has an independent positive or negative charge.

02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter.. To know more differences download byju's the … 28/03/2011 · ions are atoms with extra electrons or missing electrons. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged.. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule.

30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule... .. An ion with more protons than electrons carries a net positive charge and is called a cation.

Ion may be composed of several atoms or a single atom. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. 28/03/2011 · ions are atoms with extra electrons or missing electrons. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ).. An ion with more protons than electrons carries a net positive charge and is called a cation.

This however contradicts what was stated earlier that they have to be electrically neutral to be stable. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. On the other hand, an ion has an independent positive or negative charge. An ion with more protons than electrons carries a net positive charge and is called a cation. When an atom's outermost orbital gains or loses electrons (also known as valence electrons ), the atom forms an ion. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. When an ion is formed, the number of. That is, they have gained or lost an electron or more to become positively or negatively charged. Ion may be composed of several atoms or a single atom. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged.

Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. While ions are charged particles, positive ions (cations) or negative ions (anions). That is, they have gained or lost an electron or more to become positively or negatively charged. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ).

When an ion is formed, the number of. While ions are charged particles, positive ions (cations) or negative ions (anions). They contain the same number of protons as electrons. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. The main difference between an atom and an ion is that the atom is electrically neutral, and an ion is electrically charged. The reason behind an atom's neutral electrical charge is the presence of an equal number of protons and electrons. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. That is, they have gained or lost an electron or more to become positively or negatively charged. 28/03/2011 · ions are atoms with extra electrons or missing electrons.

It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge.. On the other hand, an ion has an independent positive or negative charge. 28/03/2011 · ions are atoms with extra electrons or missing electrons. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). Ion may be composed of several atoms or a single atom. That is, they have gained or lost an electron or more to become positively or negatively charged. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. They contain the same number of protons as electrons. An ion with more protons than electrons carries a net positive charge and is called a cation. While ions are charged particles, positive ions (cations) or negative ions (anions). On the other hand, an ion has an independent positive or negative charge.

30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule... It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. Ion may be composed of several atoms or a single atom. 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. An ion with more protons than electrons carries a net positive charge and is called a cation. That is, they have gained or lost an electron or more to become positively or negatively charged.

Ion may be composed of several atoms or a single atom. An ion with more protons than electrons carries a net positive charge and is called a cation. While ions are charged particles, positive ions (cations) or negative ions (anions). This however contradicts what was stated earlier that they have to be electrically neutral to be stable. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ).. To know more differences download byju's the …

It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge.. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators. That is, they have gained or lost an electron or more to become positively or negatively charged. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. While ions are charged particles, positive ions (cations) or negative ions (anions). They contain the same number of protons as electrons.. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds.

30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule. Ion may be composed of several atoms or a single atom.

While ions are charged particles, positive ions (cations) or negative ions (anions).. That is, they have gained or lost an electron or more to become positively or negatively charged.. To know more differences download byju's the …

This however contradicts what was stated earlier that they have to be electrically neutral to be stable.. This however contradicts what was stated earlier that they have to be electrically neutral to be stable. 09/04/2010 · about press copyright contact us creators advertise developers terms privacy policy & safety how youtube works test new features press copyright contact us creators.. Ion may be composed of several atoms or a single atom.

This however contradicts what was stated earlier that they have to be electrically neutral to be stable. To know more differences download byju's the …
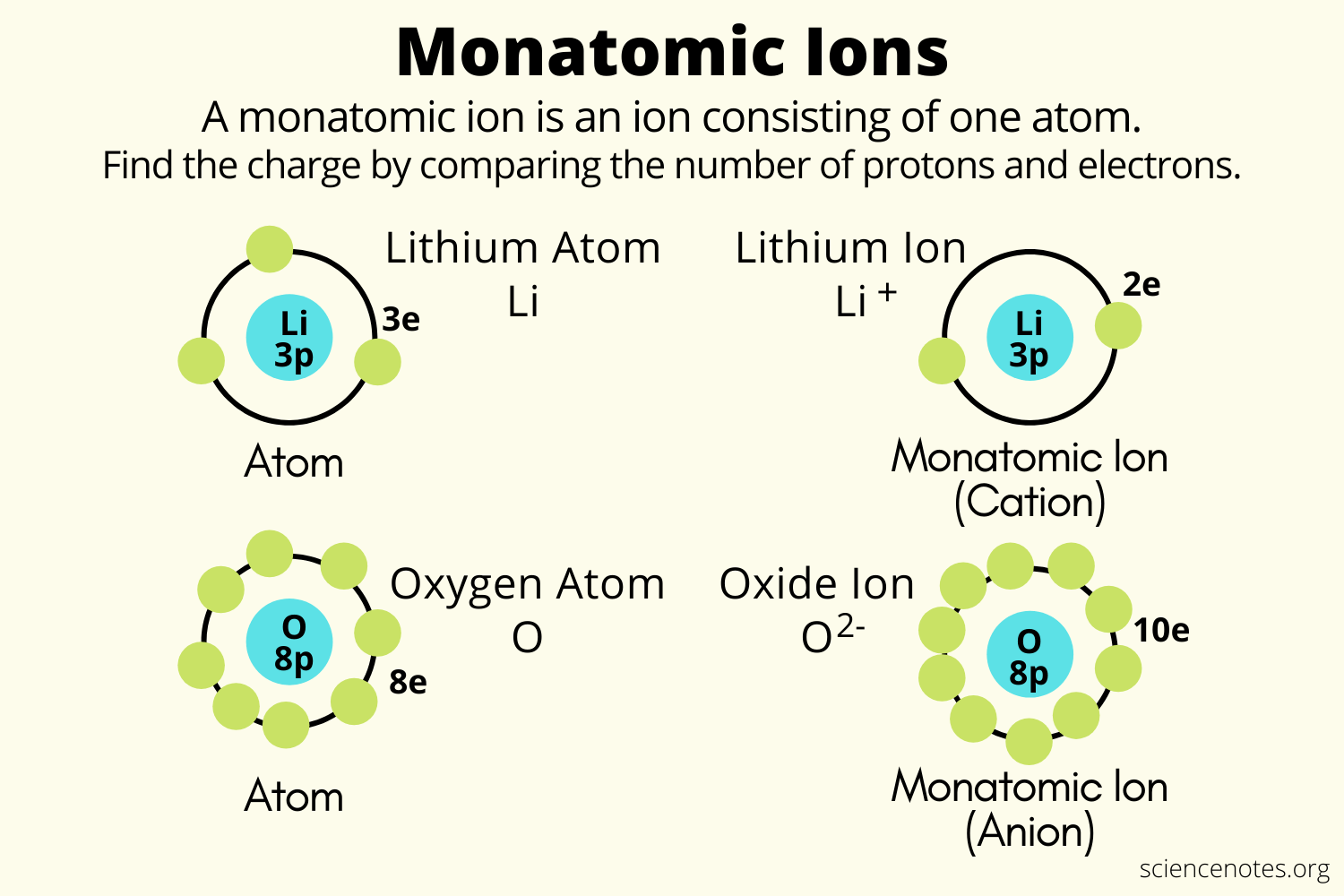
Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds.. Atoms are unique and do not differentiate into various types while ions have two types as positive ions ( cations) and negative ions ( anions ). It contains the equal number of electrons and protons in their nucleus, thus bear a net neutral electrical charge, whereas ion is an atom that contains the unequal number of protons and electrons in it and therefore carries a net positive or negative charge. They contain the same number of protons as electrons. While ions are charged particles, positive ions (cations) or negative ions (anions). 02/02/2020 · the main difference between atom and ion is that atom is the fundamental unit of matter. Combination of atoms results in the formation of molecules and the combination of ions results in the formation of compounds. To know more differences download byju's the … That is, they have gained or lost an electron or more to become positively or negatively charged. An ion with more protons than electrons carries a net positive charge and is called a cation.. 30/06/2011 · atom is the smallest neutral unit which can form bonds while ion is any charged molecule.